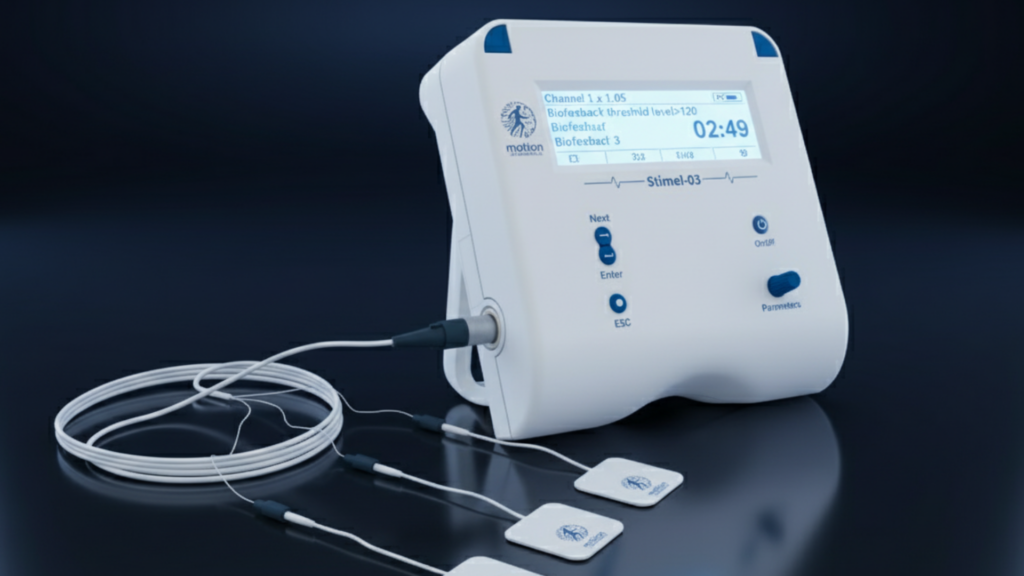
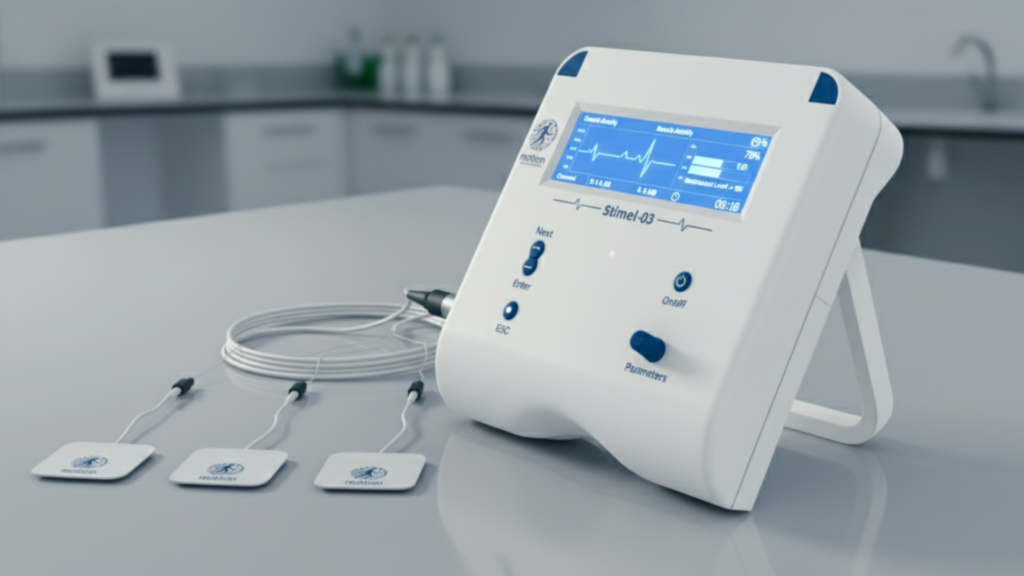
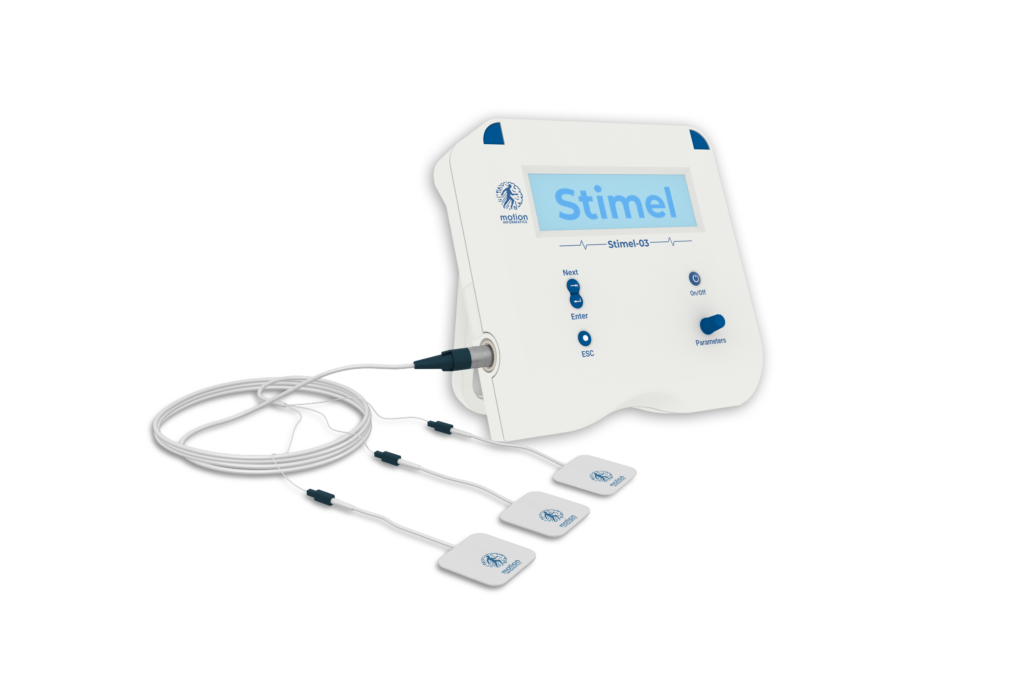
STIMEL-03: Accelerating Recovery with Tailored Stimulation
An FDA-cleared, EMG-driven neuromuscular stimulation system integrating Functional Electrical Stimulation (FES), Neuromuscular Electrical Stimulation (NMES), and real-time biofeedback into a single adaptive rehabilitation solution. Powered by BioRhythmIQ technology, Stimel-03 captures electromyographic signals, analyzes neuromotor intent in real time, and delivers precisely timed stimulation to reinforce correct motor activation patterns.
-
Accelerated Motor Recovery
EMG-triggered stimulation reinforces voluntary intent in real time, driving faster functional gains and measurable improvement in activities of daily living.
-
Active Neuroplasticity Engagement
Synchronization of brain intent, visual biofeedback, and electrical stimulation strengthens motor pathways and supports durable neural rewiring.
-
Personalized Patient-Specific Therapy
Individual EMG thresholds and adaptive stimulation parameters reduce fatigue, prevent over-stimulation, and align therapy with each patient\\\\\\\'s neuromuscular status.
-
Early & Chronic Stage Applicability
High-sensitivity EMG detection enables intervention even when visible movement is minimal, supporting early post-stroke care and long-term rehabilitation.
Why Stimel-03
Key Differentiators
-
Intent-Triggered Stimulation
Stimulation fires only when the patient\\\\\\\'s own muscle signal is detected - reinforcing voluntary effort rather than replacing it.
-
Real-Time Timing Alignment
Stimulation is synchronized with neuromotor intent, not delivered on a fixed schedule.
-
Adaptive Threshold Modulation
The system continuously adjusts EMG detection thresholds to account for muscle fatigue and changing baseline activity.
-
Multi-Channel Precision
Up to 8 independent stimulation channels allow targeted activation of specific muscle groups simultaneously.

Precision Technology
Adaptive Biofeedback
BioRhythmIQ technology captures patient-specific electromyographic signals and displays them in real time. The dynamic EMG threshold algorithm recalibrates continuously based on performance and fatigue. once the patient’s muscle activation reaches a dynamically calibrated threshold, stimulation fires to reinforce correct motor execution. Passive stimulation becomes active neuromuscular re-education.
- EMG-triggered stimulation with <8ms latency
- Adaptive threshold modulation prevents over-stimulation
- Real-time visual and auditory biofeedback
- Session data logged for clinician review
Autonomous Recovery
Clinical Results
Stimel-03 delivers measurable clinical outcomes: ADL improvement (FIM) of +50.6% vs +20.7% in controls; hand function (Fugl-Meyer) +67.4% vs +34.8%; stroke severity (NIHSS) -48.7% vs -26.2%. Recovery is 2.3x faster than controls, with fewer sessions. The system detects ultra-low EMG signals, enabling early intervention even when visible movement is minimal.
- Stimulation Accuracy
- 94%
- Trigger Latency
- <8ms
- Battery Life
- 12hrs
Technical Specifications
Standard performance metrics for clinical evaluation.
| Parameter | Value | Notes |
|---|---|---|
| Stimulation Channels | Up to 8 | Independent multi-channel output |
| EMG Sample Rate | 2,000 Hz | Real-time intent detection |
| Pulse Width | 20–500 μs | Programmable per channel |
| Max Output Current | 120 mA | Per channel, adjustable |
| Battery Life | Up to 12 hours | Full clinical session |
| Connectivity | BLE 5.2 / USB-C | Wireless and wired sync |
Product Demonstration
See STIMEL-03® in Action
Watch real clinical demonstrations of the STIMEL-03® neuromuscular stimulation system delivering precise, intent-triggered therapy.

Ready to transform patient recovery?
Join hundreds of clinical centers worldwide already utilizing the STIMEL-03 system.

